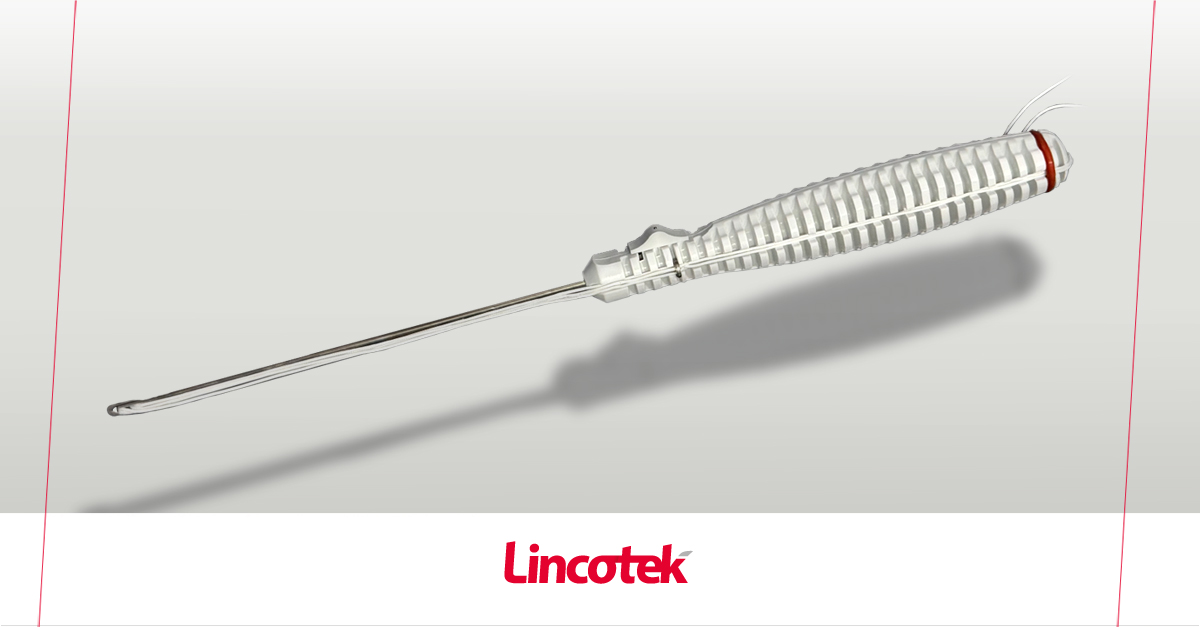
Ankle Stabilization Device now available for private labeling
Memphis, OH (US) | April 20, 2026: Lincotek is proud to announce that it has recently received 510(k) clearance (K252081) from the U.S. Food and Drug Administration (FDA) for its SportLinc™ Syndesmosis Device, a sterile, single-use implant designed to stabilize syndesmotic trauma of the ankle.
The implant features an ultra-high-molecular-weight polyethylene (UHMWPE) suture, tensioned between two low-profile titanium alloy buttons. It can be used alongside plate fixation or as a standalone solution, offering flexibility in ankle stabilization. The preloaded button-suture assembly is provided in a sterile inserter instrument, supporting ease of use in the operating room.
As a single-use system including drill, drill guide and tension handles, SportLinc™ is designed to meet the needs of both hospitals and Ambulatory Surgery Centers (ASCs), helping streamline procedures and reduce operational complexity.
The device is commonly used in sports trauma and ankle fractures. A syndesmosis injury (commonly referred to as a high ankle sprain) damages the ligaments connecting the tibia and fibula above the ankle, often caused by forced external rotation of the foot. SportLinc™ is designed to provide support to the injury throughout the healing process.
The product is available for private labeling and can also serve as a predicate device, enabling OEM partners to accelerate the development and commercialization of similar solutions in the trauma and sports medicine markets.
Francesco Bucciotti, Head of Global Business and Business Development at Lincotek’s Medical Division, said: “We pride ourselves on helping customers bring products to market in less than one year from the design concept to the 510k approval, keeping them ahead in an increasingly competitive industry. SportLinc™ has big commercial potential and will have a strong appeal to clinicians dealing with sports injuries. With FDA clearance in place, we’re now ready to provide a market-ready solution to orthopedic OEMs.”
Product Development Director, Troy Walters, added: “SportLinc™ was developed with a focus on balancing strength, flexibility, and ease of use – three elements that are essential in modern orthopedic surgery. It reflects the expertise and dedication of the Lincotek product development team in supporting orthopedic OEMs with continuously innovative solutions.”
Lincotek, which has a long history of innovation and product development in the field of medical devices, will work towards a CE Mark to complement the 510k approval in the United States, and with a view to bringing SportLinc™ to European markets.



